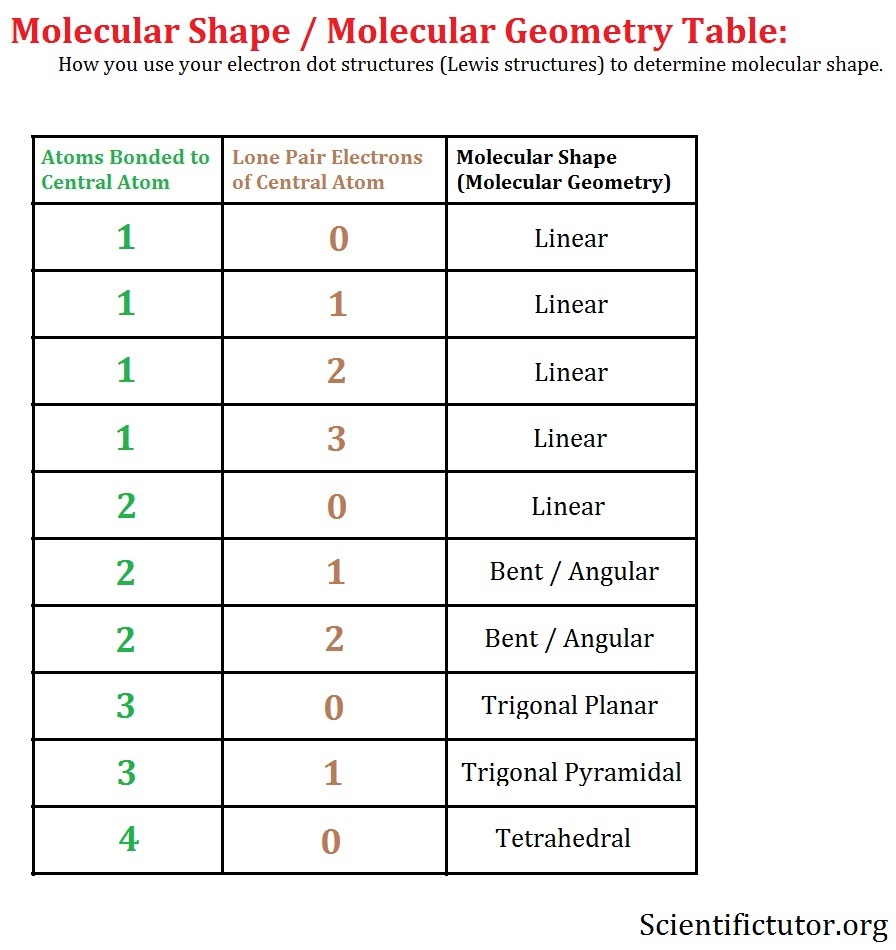 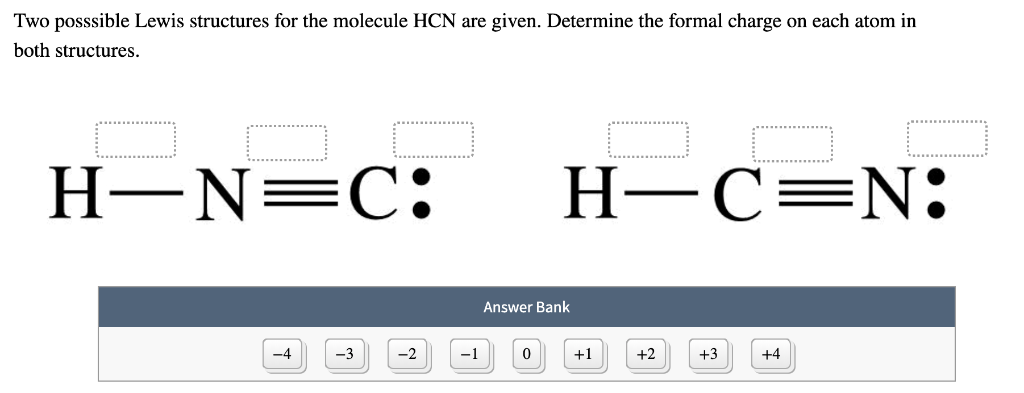
If any atoms lack an octet, make a double or triple bond to give them an octet.Add the remaining electrons to satisfy the octet for a more electronegative atom first.Use a pair of electrons to form a bond between each pair of bound atoms.Sum the valence electrons from all the atoms.* Put more electronegative elements in terminal positions * Hydrogen atoms are always terminal (only one bond) Write the correct skeletal structure for the molecule.Determine the name of the electron and molecular geometry.Arrange them in a way that minimizes repulsion (as far apart as possible).Count the number of atoms and lone pairs of electrons on the central atom ( steric number).Determine the central atom and draw the Lewis structure for the molecule.The B–B bond length is 1.76 Å.Follow these steps to determine the electron and molecular geometries: In the tenth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the ninth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. 
In the eighth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There is one shorter (1.76 Å) and one longer (1.77 Å) B–B bond length. In the seventh B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. There is one shorter (1.75 Å) and one longer (1.79 Å) B–B bond length. In the sixth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fifth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fourth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the third B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the second B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the first B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There are a spread of Mg–B bond distances ranging from 2.48–2.52 Å. In the fifth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.47–2.51 Å. In the fourth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.48–2.51 Å. In the third Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.49–2.51 Å. In the second Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with three equivalent LiB12 cuboctahedra, edges with nine MgB12 cuboctahedra, and faces with eight MgB12 cuboctahedra. There are a spread of more » Mg–B bond distances ranging from 2.48–2.50 Å. In the first Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra, faces with two equivalent LiB12 cuboctahedra, and faces with six MgB12 cuboctahedra. There are a spread of Li–B bond distances ranging from 2.48–2.50 Å. Li is bonded to twelve B atoms to form LiB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra and faces with eight MgB12 cuboctahedra. LiMg9B20 is hexagonal omega structure-derived structured and crystallizes in the triclinic P-1 space group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
